An Omicron Vaccine Is Being Tested Now — Here’s What You Need To Know
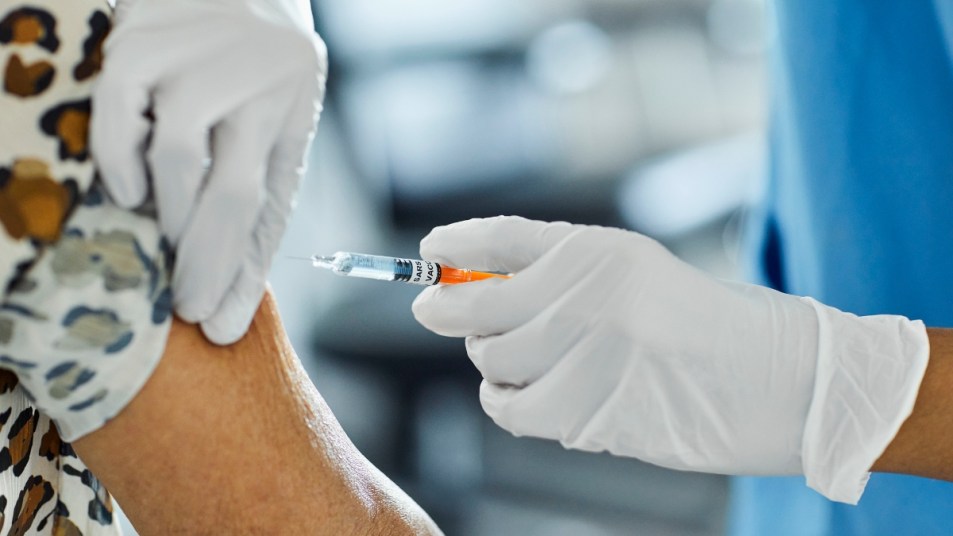
While it seems that the current COVID-19 vaccines are here to stay, researchers haven’t stopped working on vaccine updates and improvements to help keep us safe from this ever-evolving virus. On Tuesday, Pfizer and BioNTech announced the beginning of a new study on an Omicron vaccine.
Researchers recruited 1,420 healthy adults, who will be some of the first people to receive the new vaccine. The clinical trial will test the safety and tolerability of the vaccine, as well as the level of immune response in the participants.
How will the clinical trial work?
The research team is testing the Omicron vaccine on participants with varying vaccination statuses. As a result, they are studying three main groups of participants, all of whom are ages 18 through 55:
- The first group includes 615 participants who have already received two doses of the current Pfizer-BioNTech COVID-19 vaccine. They had both shots 90 to 180 days prior to enrolling in the study, and the researchers will give them either one or two doses of the new Omicron vaccine.
- Participants in the second group (600 total) have already received three doses of the current Pfizer vaccine, and had their last shot 90 to 180 days prior to enrollment. This group will receive either one dose of the current Pfizer vaccine or one dose of the Omicron vaccine.
- The third group, which consists of 205 participants, is made up of people who haven’t received any COVID-19 vaccines. The researchers will give these volunteers three doses of the Omicron vaccine.
Why do we need an Omicron vaccine?
It’s clear that the current Pfizer and Moderna vaccines are effective. Two doses protect against serious infection, and a booster dose protects us even further against infection and severe illness. However, early evidence suggests that the vaccines on the market are less effective against Omicron.
“While current research and real-world data show that boosters continue to provide a high level of protection against severe disease and hospitalization with Omicron, we recognize the need to be prepared in the event this protection wanes over time and to potentially help address Omicron and new variants in the future,” said Kathrin U. Jansen, PhD, Senior Vice President, and Head of Vaccine Research and Development at Pfizer. “Staying vigilant against the virus requires us to identify new approaches for people to maintain a high level of protection, and we believe developing and investigating variant-based vaccines, like this one, are essential in our efforts towards this goal.”
It will take some time to see the results of this study, but it’s an important step forward in the race against COVID-19.













