FDA Approves First-Ever Diabetes Test That Doesn’t Require a Fingerstick
For those who suffer from type 1 or type 2 diabetes, the journey can be painful and frustrating: adhering to a strict diabetes diet, trying out and taking different diabetes medications, and daily diabetes tests that require a finger stick (pricking the fingertip for a sample of blood.) There is now new technology for those who are tired of pricking their finger multiple times a day to measure their glucose levels.
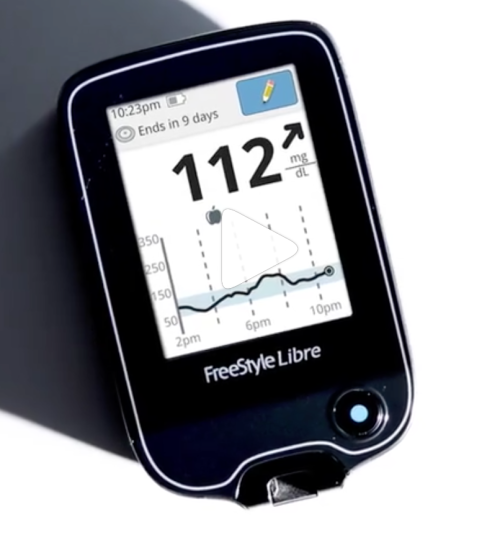
The Food and Drug Administration has just approved the first ever diabetes test kit that doesn’t require a blood sample from the finger tip. FreeStyle Libre Flash Glucose Monitoring System, made by Abbott Diabetes Care, Inc., is the device that could change how people with diabetes live their lives.
The system uses a small censor wire that continuously monitors glucose levels. A mobile reader is waved above the censor to measure glucose levels, and to check if glucose levels are too high, hyperglycemia, or if glucose levels are too low, hypoglycemia.
“The FDA is always interested in new technologies that can help make the care of people living with chronic conditions, such as diabetes, easier and more manageable,” said Donald St. Pierre, deputy director of new product evaluation in the FDA’s Center for Devices and Radiological Health in a release. “This system allows people with diabetes to avoid the additional step of fingerstick calibration, which can sometimes be painful, but still provides necessary information for treating their diabetes — with a wave of the mobile reader.”
The device is applied to the back of the arm by a medical professional, and is for people 18 years or older. The censor can be worn for up to 14 days. After two weeks, the patient returns to their doctor’s office to have their glucose reports downloaded. There are several side effects with the device, (as there is with all medications) including skin irritation around the implant, inaccurate readings, and the inability to alert glucose changes without the user initiating a reading.
According to the Centers for Disease Control and Prevention, more than 30 million people in the United States suffer from diabetes. We hope this device makes their day-to-day lives a bit easier.
h/t Fortune
More from Woman’s World
6 Ways to Control Your Blood Sugar Levels to Prevent Diabetes and Cancer
The Glucose Monitor Said Sick Little Boy’s Blood Sugar Was Normal, But Faithful Dog Discovered Something Child’s Own Mother Couldn’t













